Events & Promotions
|
|

GMAT Club Daily Prep
Thank you for using the timer - this advanced tool can estimate your performance and suggest more practice questions. We have subscribed you to Daily Prep Questions via email.
Customized
for You
Track
Your Progress
Practice
Pays
Not interested in getting valuable practice questions and articles delivered to your email? No problem, unsubscribe here.
- Jun 08
08:00 PM EDT
-10:00 PM EDT
Master the GMAT with expert live instruction, a personalized study plan, and real-time support. Includes 40 hours of online classes plus 6 months of access to the TTP GMAT OnDemand video course. Mon/Wed June 8, 2026 →August 12, 2026 8:00pm-10:00pm EST - Jun 10
06:00 AM PDT
-06:15 PM PDT
Register for the GMAT Club Virtual MBA Spotlight Fair – the world’s premier event for serious MBA candidates. This is your chance to hear directly from Admissions Directors at nearly every Top 30 MBA program.. - Jun 10
10:00 AM PDT
-11:00 AM PDT
Scoring 715 on the GMAT Focus Edition requires more than just learning formulas, memorizing concepts, or solving hundreds of questions. In this episode, Nishant shares how he improved his GMAT preparation by focusing on application of concepts, and more. - Jun 11
11:00 AM EDT
-01:00 PM EDT
TTP GMAT OnDemand gives serious students 400+ hours of expert video instruction, the full TTP course, AI support, weekly office hours, and a 715+ score guarantee—all built for elite GMAT score improvement.
Originally posted by Sajjad1994 on 09 Feb 2021, 07:30.
Last edited by BottomJee on 03 Dec 2023, 00:56, edited 2 times in total.
Last edited by BottomJee on 03 Dec 2023, 00:56, edited 2 times in total.
Moved to new forum
Kudos
Bookmarks
Dropdown 1: Radio Waves << Infrared << Visible Light << X-Rays << Gamma Rays
Dropdown 2: No, he cannot
Be sure to select an answer first to save it in the Error Log before revealing the correct answer (OA)!
Difficulty:
 55%
(hard)
55%
(hard)
Question Stats:
62% (01:52) correct 38%
(02:01)
wrong
38%
(02:01)
wrong  based on 1940
sessions
based on 1940
sessions
History
Date
Time
Result
Not Attempted Yet
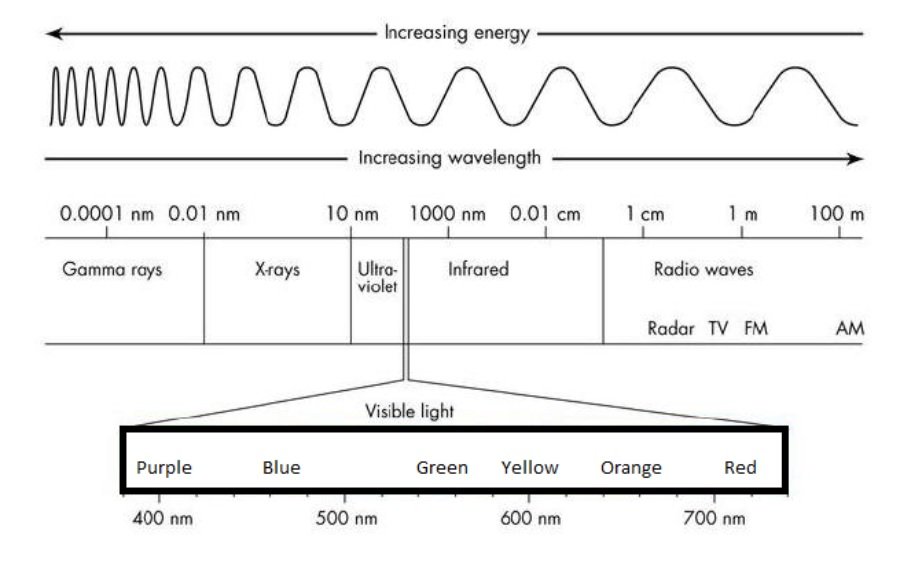
The diagram below shows the electromagnetic spectrum arranged by wavelength and energy level.
 Based on the diagram, answer the questions that follow.
Based on the diagram, answer the questions that follow.
1. Which of the following sequences properly orders its types of electromagnetic radiation by amount of energy?
2. A scientist is conducting an experiment and finds out that the electromagnetic radiation he is using has a wavelength of 573 cm. Can he see the radiation without other special equipment?
3.jpg [ 81.82 KiB | Viewed 13462 times ]
1. Which of the following sequences properly orders its types of electromagnetic radiation by amount of energy?
2. A scientist is conducting an experiment and finds out that the electromagnetic radiation he is using has a wavelength of 573 cm. Can he see the radiation without other special equipment?
Attachment:
3.jpg [ 81.82 KiB | Viewed 13462 times ]
ShowHide Answer
Official Answer
Dropdown 1: Radio Waves << Infrared << Visible Light << X-Rays << Gamma Rays
Dropdown 2: No, he cannot
rocky620

Retired Moderator
Joined: 10 Nov 2018
Last visit: 11 May 2023
Posts: 479
Given Kudos: 229
Location: India
Concentration: General Management, Strategy
Schools: IIMA PGPX'22 IIMB EPGP'22 IIMC PGPEX'22 ISB'22
GMAT 1: 590 Q49 V22

WE:Other (Retail: E-commerce)
Kudos
Bookmarks
1.1 Which of the following sequences properly orders its types of electromagnetic radiation by amount of energy?
The radiated energy levels are increasing from Right to Left
The order is - Radio Waves < Infrared < Visible Light < Ultra Violet < X-Rays < Gamma Rays
Only Option D matches this sequence.
A. Gamma Rays <<Ultraviolet << Visible Light << X-Rays <<Infrared
B. Gamma Rays << X-Rays << Visible Light Infrared << Radio Waves
C. Radio Waves << Infrared << Ultraviolet << Gamma Rays << X-Rays
D. Radio Waves << Infrared << Visible Light << X-Rays << Gamma Rays
1.2 A scientist is conducting an experiment and finds out that the electromagnetic radiation he is using has a wavelength of 573 cm. Can he see the radiation without other special equipment?
573 cm = 5.73 meters
Visible light falls in the wavelength of 380 nm - 740 nm. So the scientist will not be able to see the radiation without any special equipment.
A. Yes, he sees green
B. Yes, he sees yellow
C. No, he cannot
1.3 Which of the following has the greatest wavelength?
Wavelengths of
A. Yellow visible light ~ 600 nm
B. Red visible light ~ 720 nm
C. Infrared radiation ~ 1000 nm
The radiated energy levels are increasing from Right to Left
The order is - Radio Waves < Infrared < Visible Light < Ultra Violet < X-Rays < Gamma Rays
Only Option D matches this sequence.
A. Gamma Rays <<Ultraviolet << Visible Light << X-Rays <<Infrared
B. Gamma Rays << X-Rays << Visible Light Infrared << Radio Waves
C. Radio Waves << Infrared << Ultraviolet << Gamma Rays << X-Rays
D. Radio Waves << Infrared << Visible Light << X-Rays << Gamma Rays
1.2 A scientist is conducting an experiment and finds out that the electromagnetic radiation he is using has a wavelength of 573 cm. Can he see the radiation without other special equipment?
573 cm = 5.73 meters
Visible light falls in the wavelength of 380 nm - 740 nm. So the scientist will not be able to see the radiation without any special equipment.
A. Yes, he sees green
B. Yes, he sees yellow
C. No, he cannot
1.3 Which of the following has the greatest wavelength?
Wavelengths of
A. Yellow visible light ~ 600 nm
B. Red visible light ~ 720 nm
C. Infrared radiation ~ 1000 nm
Kudos
Bookmarks
can someone please help me understand the 2nd part?











